As a first assistant in the maternity ward, Mary Chiesa watched in horror when a mother lost grip of her newborn shortly after a C-section. The baby fell to the hard operating room floor when the mother’s blood pressure cuff inflated, opening her arm enough for the child to slip out. The baby was whisked away, and due to privacy laws, Chiesa never heard more about the baby. It has haunted her ever since.
“When the nurses and other teams were discussing it later, I was surprised to learn this wasn’t uncommon,” Chiesa said. “In fact, more than 1,600 babies are reported dropped in the first days of life. And it’s believed only about one third are actually reported. I felt on a deep level I had to do something.”
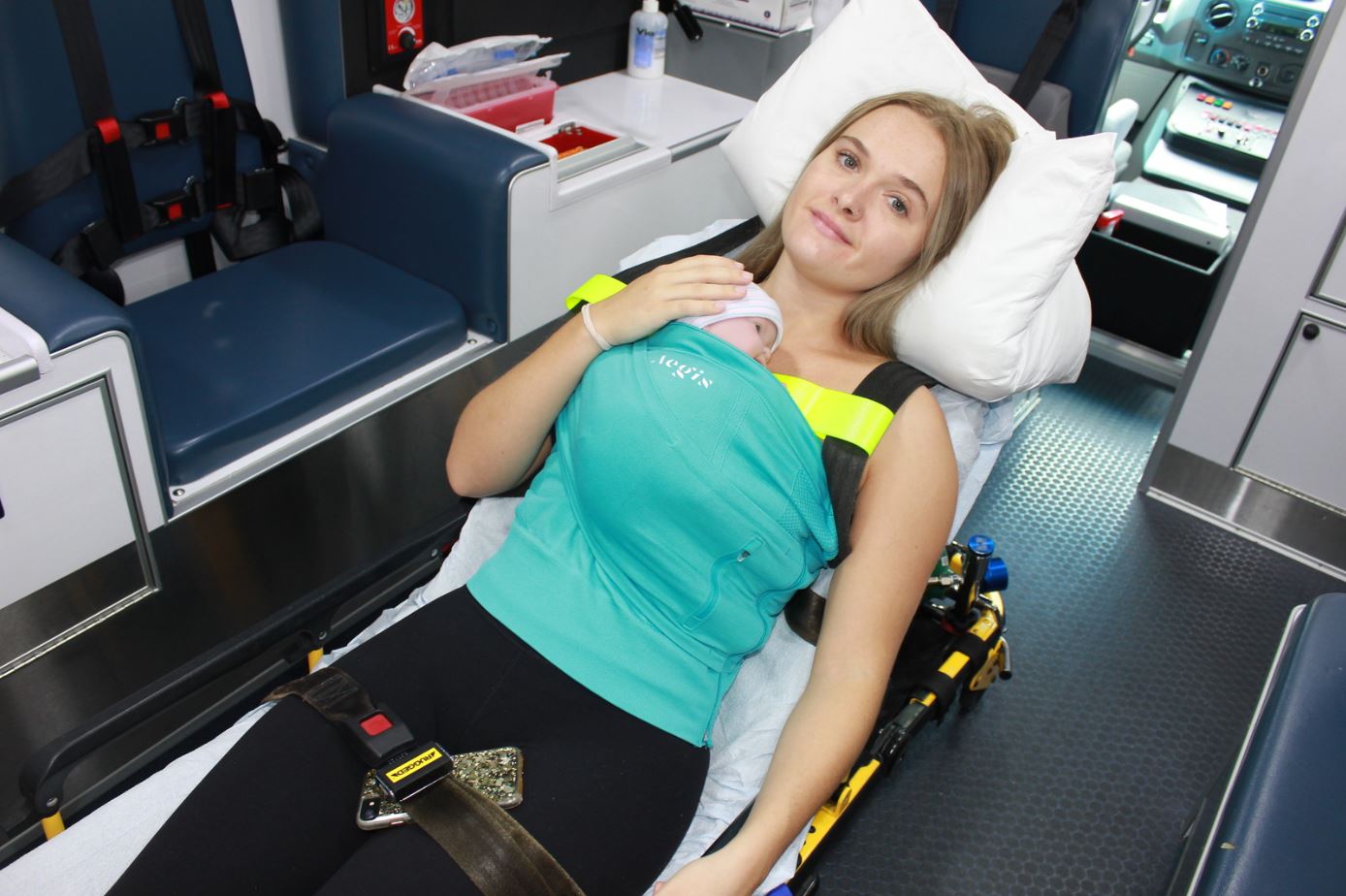
Not long after the incident, Chiesa founded Saplacor and began designing and testing a solution: the Aegis Neonate Medical Wrap. Originally designed to comfortably and securely hold baby close to mom, dad, or other caregiver in the hospital while leaving the adult’s hands free, the Aegis quickly showed promise for medical transport.
Aegis is made of seamless four-way stretch nylon and elastin, interwoven with X-Static silver thread for thermal, antimicrobial, and anti-odor protection. The tube top style device securely holds baby in a frog leg position against an adult chest without interfering with adult transport seatbelt protection. A side locking zipper access allows for tube, line, and monitor access for mother and baby while a shoulder sling allows for mobile support. Meant to be sent home with the mother, the device is available in five adjustable sizes to accommodate a range of adult body types and is designed to hold infants from four to 14 pounds (1.8 to 6.3 kilograms).
“Transporting a newborn has always been a challenge and that’s particularly true in the air where it’s louder and colder,” Chiesa said. “Mother is restrained, and the infant is often held unrestrained or separated from mom, which increases risks of hypothermia and transport injuries. Wrapped securely with skin-to-skin contact, baby’s transport risks are greatly reduced while the benefits include keeping baby warm, reducing stress, providing access to food that is otherwise not available on an aircraft, and, of course, increased physical safety.”
Named after the Greek myth around the protective shield of Zeus and Athena, Aegis underwent significant testing before receiving designation as a Food and Drug Administration-approved general wellness device. Testing included frontal, rear, and side impact force testing; ASTM testing for strength and chemical safety; and Nobel Lab testing, where it received designation as antimicrobial and antifungal. After more than a year of research, development, and testing, Aegis hit the market in April 2018. Today, more than 1,000 have been sold to hospitals, ground transport services, and air ambulance companies.
“Maternity is the only time a patient is expected to care for another patient,” Chiesa said. “Mom and baby need skin-to-skin contact for post-partum recovery. With blood pressure cuffs, IVs, medication, and a host of other things happening to mom, and sometimes baby too, there was a real need for a secure way to secure baby in transport to take advantage of those skin-to-skin benefits of reduced stress, improved heart and lung function, stabilized body temperature, regulated blood sugar, reduced postpartum depression, colonizing good bacteria, and boosted bonding. Aegis allows babies to experience these immediately after birth even if they need transport. Moms report they keep using the Aegis for months after to keep baby safe and close every day.”
The Aegis is available online from Saplacor. Hospitals and ambulances can purchase the device for $79. The reusable ABEO pregnancy seatbelt attachment is available for $20.